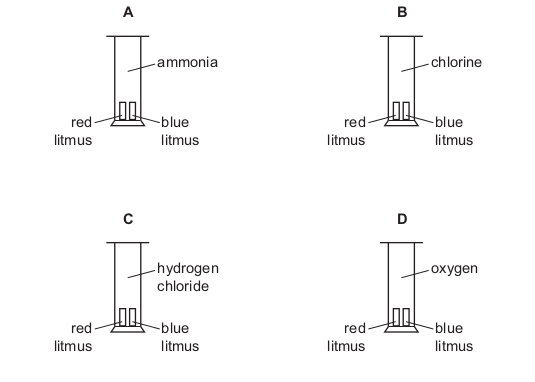
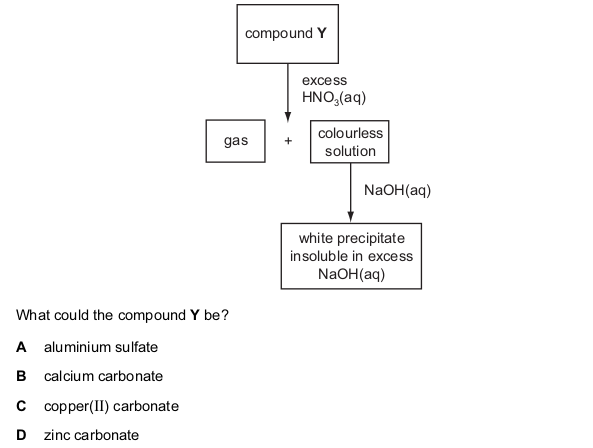
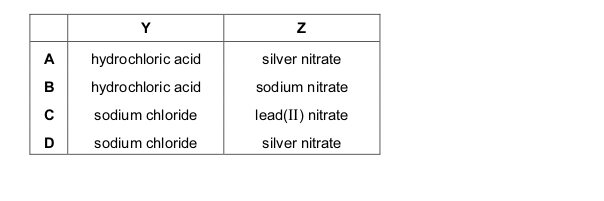
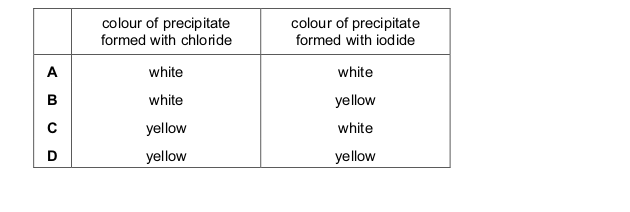
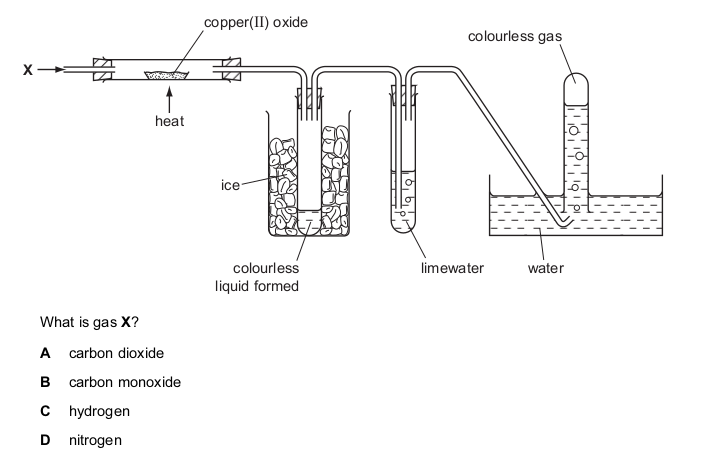
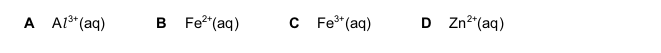One very important aspect of gcse chemistry is the identification of ions (which is an atom or molecule having a net electrical charge, this charge could be negative as in the case of an electron, positive as in the case of protons or just neutral). gcequiz.com has brought together questions in the identification of ions and gasses as seen in the gcse. Questions that are up to examination standards.
There are so many ways to test the presence of an ion in a gas or in a solution. Some of these methods include flame tests, it’s reactivity with some metal wires such as nichrome or platinum.
Apart from chemistry quizzes, other quizzes have been made available for you on this platform to ease your studies and preparations for the end of year exams.
Good luck