Question 1 of 17
Which metal is added to aluminium to make a stronger alloy?
This Extraction of Metals Quiz quiz contains 17 multiple choice questions designed to help you revise and test your Extraction of metals quizzes knowledge. Select an answer for each question and click “Submit Answer” to see instant feedback. Take your time and try to score as high as possible!
The extraction of metals in chemistry is simply the process by which metals are gotten from their ores buried deep under the ground. The metal deposits are known as metal ores which are in varying abundances in the crust of the Earth. The metal ore is always very different from the finished metals that we see in bridges and buildings.
The extraction of metals plays a very vital role in every civilisation in that the extracted metals are used to make coins, farming tools, weapons for war, building tools and even used in the construction of buildings. These metals have changed the lives of millions over the world.
Have you learned enough about Metals ? Are you confident enough to answer the quiz? To view other quizzes in gcse chemistry Click here.
Good luck
Question 1 of 17
Which metal is added to aluminium to make a stronger alloy?
Question 2 of 17
Which of these metals should react with dilute hydrochloric acid, but very slowly?

Question 3 of 17
Which metals is obtained from bauxite ore?
Question 4 of 17
Which of (1) - (5) represents where molten iron is tapped off?
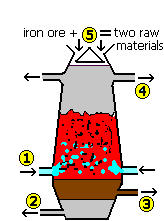
Question 5 of 17
Which metal is less reactive than sodium but must be extracted using electrolysis of the molten ore?
Question 6 of 17
Stages in the extraction of iron in a blast furnace are described below after iron ore, coke and limestone have been added at the top, but not in the correct order. (1) carbon dioxide reacts with coke (carbon) to form carbon monoxide (2) limestone combines with acid impurities (3) hot air is blown into the furnace (4) carbon monoxide reacts with iron oxide to make iron (5) coke (carbon) burns to form carbon dioxide Which is the correct order of sequence for (1) to (5)?
Question 7 of 17
Which metals is purified by electrolysis?
Question 8 of 17
Which is NOT a raw material in the blast furnace extraction of iron?
Question 9 of 17
Which reaction is causes the high temperature in a blast furnace?
Question 10 of 17
Which chemical change corresponds to a neutralisation?
Question 11 of 17
The name of a common ore from which aluminium can be extracted is called?
Question 12 of 17
The reactions in the blast furnace to produce iron are summarised by equations (1) to (4). The symbol equations may NOT be numerically balanced.
... (1) C + O2 ==> CO2
... (2) CO2 + C ==> CO
... (3) Fe2O3 + CO ==> Fe + CO2
... (4) CaCO3 + SiO2 ==> CaSiO3 + CO2
Which statement is TRUE?
Question 13 of 17
Which reaction represents the iron ore reduction in a blast furnace?
Question 14 of 17
Ingots of aluminium from its electrolytic extraction can be stored outside in all weathers. Why do aluminium ingots not corrode away, despite aluminium being quite high in the reactivity series of metals?
Question 15 of 17
Which removes impurities from iron in the blast furnace extraction of iron?
Question 16 of 17
The reactions in the blast furnace to produce iron are summarised by equations (1) to (4). The symbol equations may NOT be numerically balanced.
... (1) C + O2 ==> CO2
... (2) CO2 + C ==> CO
... (3) Fe2O3 + CO ==> Fe + CO2
... (4) CaCO3 + SiO2 ==> CaSiO3 + CO2
Which statement is TRUE?
Question 17 of 17
Which of these blast furnace reactions forms the 'slag'?