Question 1 of 20
Regions in a thermometer defined or specified and clearly separated by the stated/standard point
This Energetics 1 Quiz ph quiz contains 20 multiple choice questions designed to help you revise and test your A level Physics Energetics Quizzes knowledge. Select an answer for each question and click “Submit Answer” to see instant feedback. Take your time and try to score as high as possible!
In this quiz on Energetics, we shall be more focused on thermometry and heat capacity. Here, we shall be looking at the different types of thermometers, measurements of temperature, and calculations on heat capacities. With this, one should be able to tackle any questions on this aspect.
Energetics is a branch of physics that studies the various forms of energy, and their transformation. Real-life usage of energy can be seen as using the electrical energy stored in batteries to power and operate our electronic gadgets, and this energy must have been converted from another form (like solar energy). Another form of energy conversion could be the conversion of chemical energy stored in food, to mechanical energy when we exercise our bodies. So what is energy?
Energy is the ability or capacity to do work. There are many kinds of energy like solar energy, electrical energy, mechanical energy, nuclear energy, etc
This quiz is worth playing. Go ahead and take the quiz, alongside other quizzes on A level Physics that are available on this platform. Good luck!
Question 1 of 20
Regions in a thermometer defined or specified and clearly separated by the stated/standard point
Question 2 of 20
Consider the equation ΔQ = ΔU + ΔW where the symbols represent their usual meanings. ΔU represents
Question 3 of 20
The Gradient of a cooling curve at a point p represents:
Question 4 of 20
A hollow metal cylinder of mass 0.5kg is heated electrically by a 12W heater in a room at 15-degree Celsius cylinder temperature rises uniformly to 25 degree Celsius in 5 minutes and later becomes constant at 45 degree Celsius.
What is the rate of loss of heat to the surroundings at the constant temperature of 45 degree Celsius.
Question 5 of 20
A hollow metal cylinder of mass 0.5kg is heated electrically by a 12W heater in a room at 15-degree Celsius cylinder temperature rises uniformly to 25 degree Celsius in 5 minutes and later becomes constant at 45 degree Celsius.
Assuming the rate of heat loss is proportional to the excess temperature over the surroundings. What is the rate of loss at 20 degree Celsius.
Question 6 of 20
What us the specific heat capacity of the metal, taking into account the loss of heat to the surrounding
Question 7 of 20
Which of these is NOT a characteristic of a good thermometer?
Question 8 of 20
Why do most liquid -in-glass thermometers use mercury as the thermometer liquid?
Question 9 of 20
Which of these processes is closely associated with the formula ΔQ = mass x S.H.C x temp rise
Question 10 of 20
One of the terms can be used to answer the question below:
Work done = current x p.d x time
Question 11 of 20
One of the above terms can be used to find the total of p.e plus k.e of its particles or system.
Question 12 of 20
The process which takes place in such a way that no heat enters or leaves the system
Question 13 of 20
For isothermal process:
Question 14 of 20
A gas is compressed by a forced that does 20J of work on the gas. It’s then allowed to cool down when 15J of energy is conducted away. What is the internal energy change of the gas
Question 15 of 20
Which of these principles utilizes the three methods of heat transfer?
Question 16 of 20
There is a law in physics which states “Heat will not flow” up a thermometer hill” unless mechanical work is expended to force it to do so “. This point
Question 17 of 20
The S.I Unit conductivity, K, is
Question 18 of 20
A closed metal vessel contains water at 30 degree Celsius. The vessel has a surface area of 0.5m square and a uniform thickness of 4mm. if the outside temperature is 15 degree Celsius. What is the loss per minute by conduction? (Thermal conductivity of metal, k=400Wm-1K-1
Question 19 of 20
What is the combined resistance 0f 12Ω and 4Ω resistors?
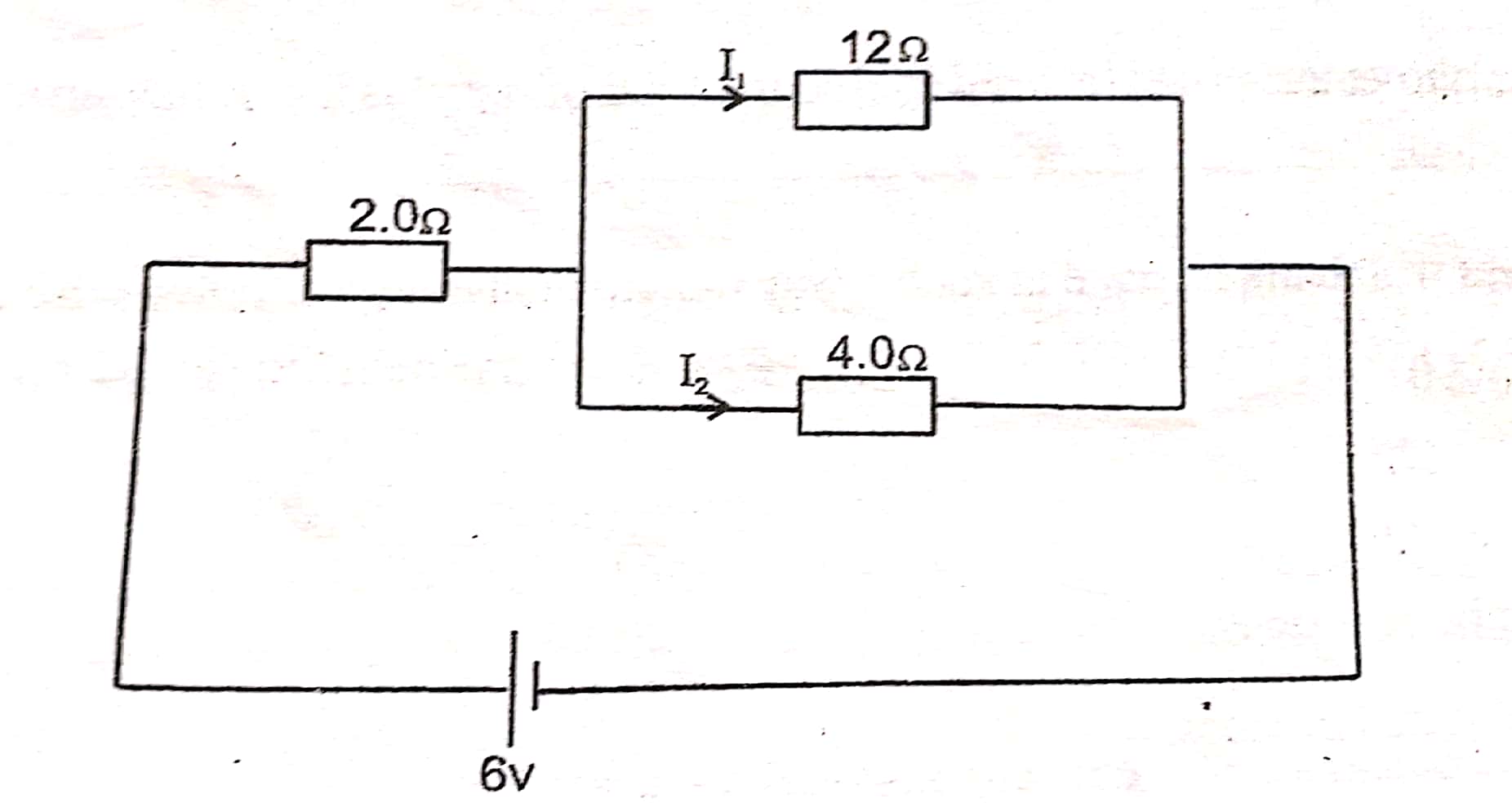
Question 20 of 20
What is the total resistance of the above circuit?
